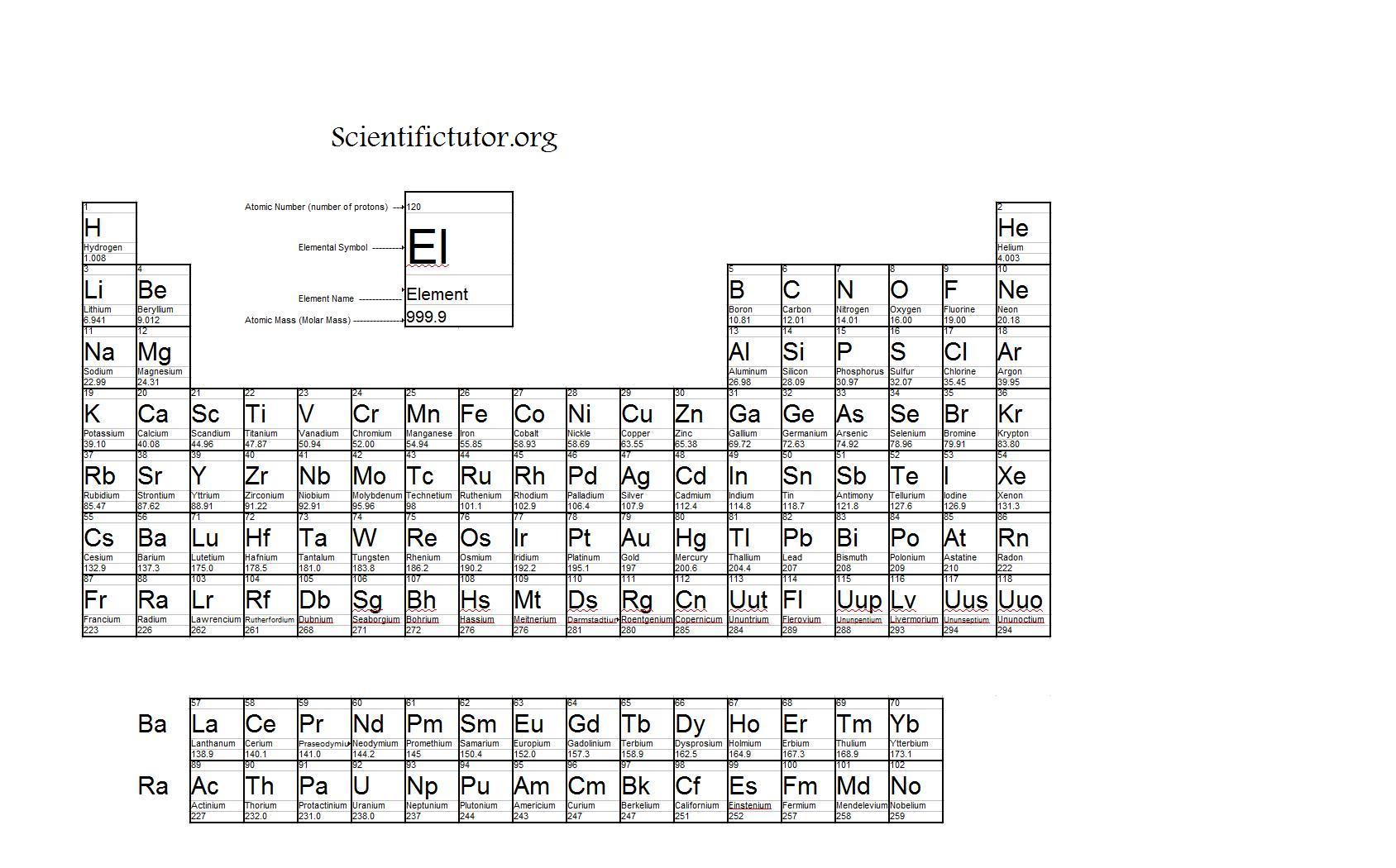
Reading Compounds that have (parenthesis's). If there is only 1 atom we do not use subscripts. The subscript belongs to the element it follows and indicates how many of that atom there are. Reading Simple Chemical Formulas- Each element begins with a CAPITAL letter. The units are atomic mass units ( amu ).ġamu =1.660 538 782×10 –27 kg 1 amu is 1/12 the mass of a carbon-12 atom which has a mass of 12.0000. Molecular Mass is the mass of a given molecule (NOT MOLES of molecules). Molecular Weight is the molar mass of a COVALENT compound. The unit for molar mass (note it is the mass of a mole) is grams/mole.Ītomic Weight is the molar mass of an element.įormula Weight is the molar mass of an IONIC compound. Molar Mass is the mass of one mole of a substance (6.02 x 10 23 formula units). As with Chemistry, the text of Wikipedia is available under the GNU Free Documentation License.(Molar Mass, Molecular Weight, Gram Formula Mass) The list of authors can be seen in the page history. 
The original article was at List of elements by atomic mass. Series: Alkalis - Alkaline earths - Lanthanides - Actinides - Transition metals - Poor metals - Metalloids - Nonmetals - Halogens - Noble gasesīlocks: s-block - p-block - d-block - f-block - g-block Name | Atomic symbol | Atomic number | Boiling point | Melting point | Density | Atomic mass Standard table | Vertical table | Table with names | Names and atomic masses (large) | Names and atomic masses (small) | Names and atomic masses (text only) | Inline F-block | Elements to 218 | Electron configurations | Metals and nonmetals | Table by blocks | Alternatives Atomic weights of elements with atomic numbers 110-116 taken from this source. IUPAC Standard Atomic Weights Revised (2005).Atomic weights of elements with atomic numbers from 1-109 taken from this source. Atomic Weights of the Elements 2001, Pure Appl.Note 5: The atomic weight of commercial Lithium can vary between 6.939 and 6.996-analysis of the specific material is necessary to find a more accurate value. 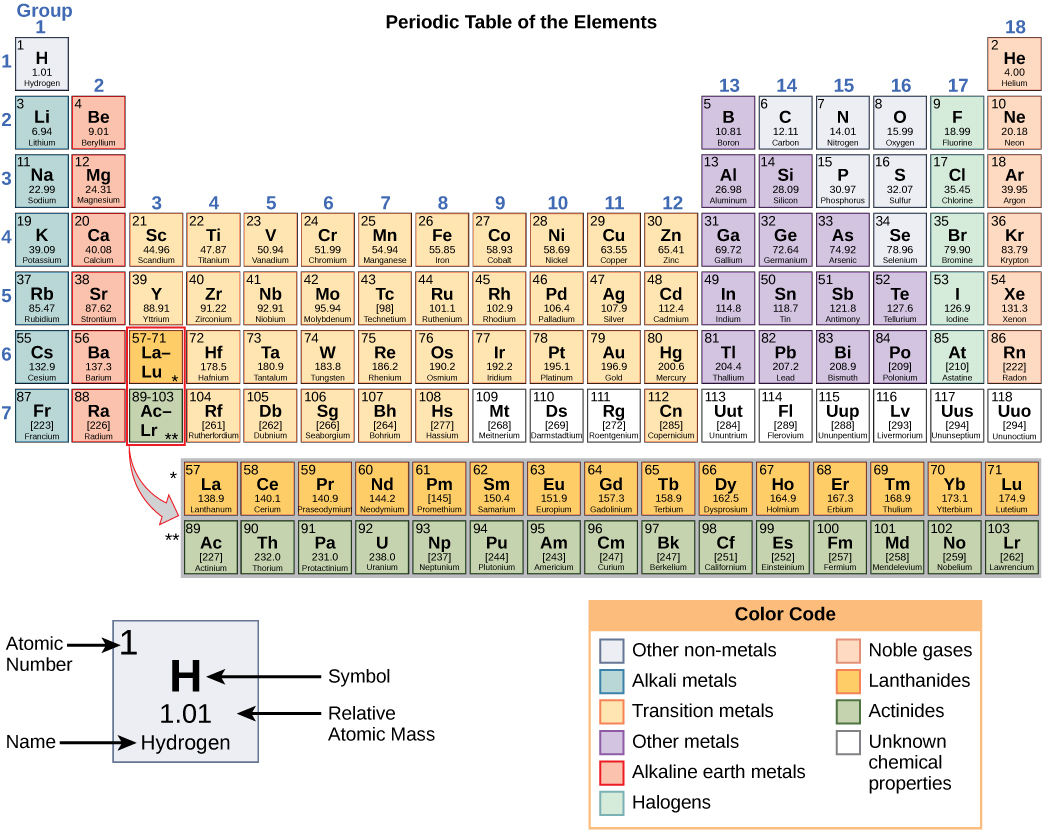
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
